Difference between revisions of "Processes & Water Filters"
| Line 71: | Line 71: | ||
== PFD Example == | == PFD Example == | ||
== Process Flow Diagram == | |||
In many engineering fields, such as chemical engineering, <b>process flow diagrams</b> (PFD) are used to indicate the sequential movement and processing of a material through multiple processing units and equipment. Drawing PFDs is an essential technique to properly and clearly relay key information about a process design. When drawing PFDs, several elements must be included in the diagram. These elements include: | |||
* Process vessels and equipment | |||
* Process and utility flow lines | |||
* Full energy and material balance | |||
* Composition of every stream | |||
* Bypass and recycle streams | |||
All processes in a PFD must abide by the law of conservation of mass and the law of conservation of energy. Energy conservation is beyond the scope of this introductory lab and will not be considered in the PFDs. Before discussing the elements of a PFD, mass conservation must be introduced. | |||
=== Problem Statement === | === Problem Statement === | ||
Revision as of 03:43, 7 September 2021
Objective
The experimental objective of this lab is to design and assemble a low-cost water filter. The contaminated water supply must first have its acidity neutralized and then filtered. The filter will be evaluated on cost and the amount of material it filters out. A process flow diagram will be designed based on the water treatment system guidelines and the physical filter design.
Overview
Access to safe drinking water is a privilege that is often taken for granted. According to the United Nations, water scarcity affects more than 40% of the global population. It is estimated that by 2050, 2.3 billion people are expected to live in areas that experience severe water stress. One of the United Nations' 17 goals to sustainably transform the world includes available and sustainable management of water and sanitation for all.
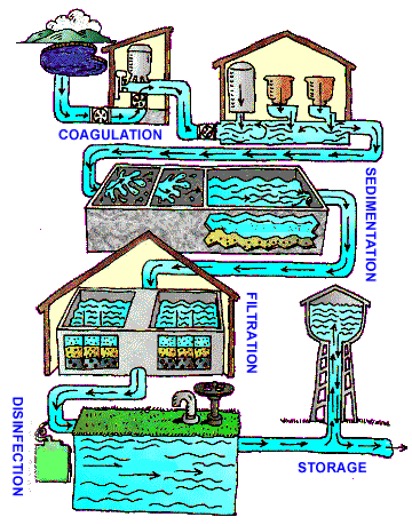
Engineers have experimented with different ways of making drinking water more accessible. Currently, water is sourced from freshwater sources, such as surface water, which includes lakes, rivers, streams, and oceans. Non-conventional water sources, such as desalinated water, treated wastewater, and agricultural drainage water, are also used. Regardless of how it is sourced, all water must go through a treatment process (Figure 1).
Water Treatment Methods
Even when water is sourced from a reservoir, it undergoes treatment to remove contaminants (Figure 2). There are five steps in the water treatment process, but this lab will use just the first three steps: coagulation and flocculation, sedimentation, and filtration.
The first step in the water treatment process is coagulation & flocculation. This is a process where positively-charged chemicals called flocculants are added to the water to neutralize the negative charge of the contaminants in the water. The contaminants combine with the flocculants to form floc, forming larger particles that are easier to filter out. In this lab, aluminum sulfate, also known as alum, is used as the flocculant.
The difference in densities between the floc and the water cause the particles to eventually settle to the bottom of the water supply. This process is sedimentation.
After sedimentation, floc is removed from the water via filtration. During filtration, the water on top of the floc goes through a series of filters that are made up of sandbeds, gravel, or other materials. The materials typically have different pore sizes to contain contaminants of different sizes. In this lab, sand, gravel, and activated carbon will be used to filter the water. At some water treatment plants, water also filters through a granulated carbon filter that removes any organic material and other particles.
For human consumption, a disinfectant may be added to kill any remaining bacteria, viruses, or parasites during disinfection. Chlorine is commonly used to treat water and rid it of any chemical pollutants. It is important that all the sediments are removed from the water as chlorine can react with them and produce trihalomethanes, which can cause cancer. Chlorine also produces a residue that will ensure water is disinfected and safe for consumption.
In the fifth step, the filtered water is stored in tanks for future use.
Water Acidity & Alkalinity
Before filtration, water must first be neutralized to alter its acidity or alkalinity. If it is not neutralized, the water may be unsafe to drink and cause corrosion of pipes in the water treatment system. Water and water-based solutions are measured for acidity or alkalinity by their power of hydrogen or potential for hydrogen (pH). The pH scale is logarithmic based on the concentration of hydrogen ions (1).
(1)
The pH of a solution indicates the concentration of hydrogen ions (H) and hydroxide ions (OH). pH is measured on a scale from 0 to 14. A pH of 7 indicates a neutral solution, which signifies that the concentration of H and OH ions is equal. If the concentration of hydrogen ions increases in a solution, it becomes more acidic. A pH of 0 to 6 indicates an acidic solution. If the concentration of hydroxide ions increases, the solution becomes more alkaline or basic. A pH of 8 to 14 indicates an alkaline solution. The pH scale and examples of solutions of varying pH values can be seen in Figure 3.
The pH of a solution can be measured using a pH strip, which is a piece of filter paper previously soaked in various acid/base indicators. Once placed in a solution, the pH strip will change color based on the acidic or alkaline nature of the solution. The pH of the solution can be approximated based on the color change of the pH strip (Figure 4).
Water Turbidity
Turbidity, which is measured in nephelometric turbidity units (NTU), is the measure of a liquid’s clarity. It is an optical characteristic of water and is found by measuring the amount of scattered light caused by materials in water. A turbidity sensor works by shining light into water and reading how much light is reflected back to it. The higher the level of turbidity, the higher the intensity of scattered light (Figure 5).
In a study on water resources conducted by Dr. Edward D. Schroeder at UC Davis, he concluded that the measure of turbidity is important regarding drinking water because microorganisms attached to contaminants are more likely to survive the disinfection process of water treatment systems.
Qualifications for Clean Water
To protect the public, the Environmental Protection Agency (EPA) sets legal limits of no more than 90 contaminants in drinking water. Additionally, secondary drinking water regulations that focus on the pH and turbidity are also set. Safe drinking water should have a pH between 6.5 and 8.5, and a standard turbidity reading of 1.0 NTU. More information about the EPA standards for clean drinking water can be found here.
Mass Balance
The law of conservation of mass states that in a closed system, the mass of the system must remain constant over time. A closed system is a type of thermodynamic system where the exchange of energy can take place with the surroundings, but the mass is conserved within the boundaries of the system. An example is a thermos, which may leak heat from the hot beverage it contains, but when properly capped does not allow the liquid to escape.
Applying the law of conservation of mass to a flow chart, the total mass that enters a system must equal the total mass exiting the same system. In Figure 5, the box represents the system, and the arrows into and out of the box represent the movement of mass into and out of the system, respectively. In a continuous process, the mass can be expressed as the mass flow rate. Mass flow rate is the mass of a fluid that passes through a cross-sectional area of the system per unit of time. In Figure 6, 7 pounds-mass of the upper material and 12 pounds-mass of the lower material enter the system per second that passes. 18 pounds-mass must leave the system per second that passes in order to satisfy mass conservation.
Mass balance can also be described with a simplified equation (2).
(2)
In (2), all the materials that are fed into the process unit are considered the input and all the product streams that flow out of the process unit are considered the output. Generation is the amount of materials formed in a process unit and consumption is the amount of materials lost in a process unit. Mass balance is also known as material balance.
PFD Example
Process Flow Diagram
In many engineering fields, such as chemical engineering, process flow diagrams (PFD) are used to indicate the sequential movement and processing of a material through multiple processing units and equipment. Drawing PFDs is an essential technique to properly and clearly relay key information about a process design. When drawing PFDs, several elements must be included in the diagram. These elements include:
- Process vessels and equipment
- Process and utility flow lines
- Full energy and material balance
- Composition of every stream
- Bypass and recycle streams
All processes in a PFD must abide by the law of conservation of mass and the law of conservation of energy. Energy conservation is beyond the scope of this introductory lab and will not be considered in the PFDs. Before discussing the elements of a PFD, mass conservation must be introduced.
Problem Statement
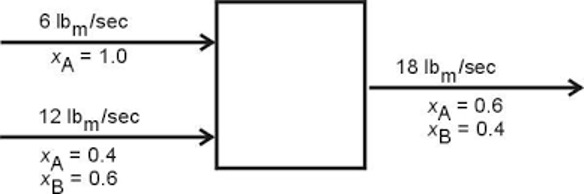
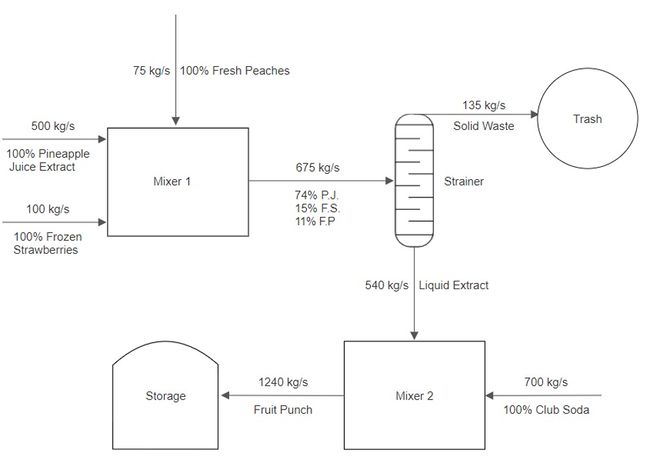
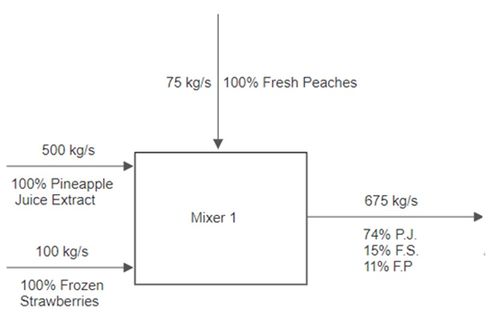
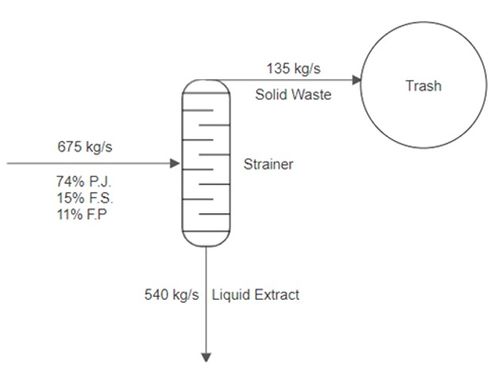
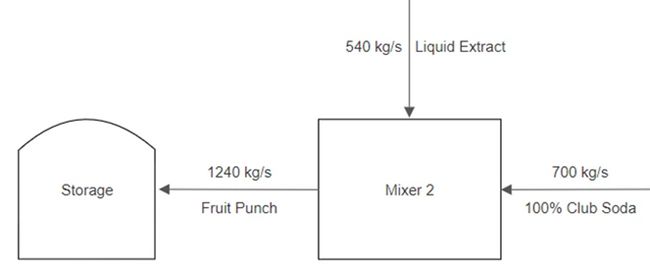
A stream of pineapple juice extract enters a mixer at a rate of 500 kg/s. A stream of frozen strawberries enters the same mixer at a rate of 100 kg/s. A stream of fresh peaches also enters the same mixer at a rate of 75 kg/s. The product stream leaves the first mixer and enters a strainer, where 80% of the mass per second exits in one stream as a liquid. Then 20% of the mass per second exits as a solid and is thrown into the trash. The liquid stream enters a final mixer alongside a stream of club soda that enters at a rate of 700 kg/s. The resulting product stream then enters a storage unit.
PFD Elements
The complete PFD in Figure 7 is used to describe the general elements of a PFD.
Process vessels and equipment are the units that the working stream passes through to have some of its physical properties changed, such as temperature, mass flow, or pressure. Some examples are mixers, steamers, turbines, and reactors. In Figure 7, the process vessels are Mixer 1, Mixer 2, Strainer, Trash, and Storage. They can be indicated by different closed form shapes.
The process and utility flow lines include the one-sided arrows in Figure 7, although a process can involve two-sided arrows. The flow lines symbolize the direction in which materials flow and the composition of each stream.
Full energy and material balance in a process involves the carrying out of the conservation of mass and energy in a process. At Mixer 1 in Figure 7, 675 kg/s enters the mixer (500 kg/s of pineapple juice, 100 kg/s of frozen strawberries, and 75 kg/s of fresh peaches), and 675 kg/s leaves the mixer. The mass and energy flow rates are usually listed above the process flow lines.
The composition of the streams is usually listed under the flow lines and indicates the percentage of the streams that each material comprises. For Mixer 1, the composition of the exit stream is indicated as 74% pineapple juice (P.J.), 15% frozen strawberries (F.S.), and 11% fresh peaches (F.P.).
Bypass and recycle streams are not considered in the PFDs in this lab, but they are also indicated by process flow lines. They consist of streams of undesirable products, such as waste products, that usually flow to a disposable unit. In Figure 7, the flow line of 135 kg/s of solid waste that flows into the trash unit is a bypass stream.
The design of a PFD based on a problem statement will be walked through in detail. The final product is illustrated in Figure 7.
Consider the first part of the example. Three streams enter a mixer and one stream leaves the mixer. There are three inputs and one output, as shown in Figure 8. Keep in mind that each stream should be labeled with the flowrate above the flow line and the composition of the stream below the flow line. Due to mass balance, the flowrate of the product stream is equal to the total flow rate of the input streams. The composition of the product stream can be calculated using (3).
(3)
To get the percentage of the concentration, multiply equation (3) by 100.
Consider the second part of the example. The product stream from Mixer 1 enters a strainer as an input, where 20% of the stream is separated into a solid stream and 80% of the stream is separated into a liquid stream. The solid stream is directed into the trash. The flowrate of the solid and liquid streams can be calculated by multiplying the percentages, 20% and 80%, by the input stream from Mixer 1, as shown in Figure 9. Due to the lack of information, the specific composition of each stream of fruit is unknown in the output and can be omitted in the PFD.
Consider the final part of the example. The liquid stream from the strainer enters a mixer as an input alongside a 700 kg/s stream of club soda. These are the input streams for the final part of the example. The product stream then enters a storage unit. Similar to the first part, the flowrate of the product stream is the sum of the input streams, as shown in Figure 10.
Design Consideration
- Which aspects of the competition ratio are most advantageous (see 2. Water Filter Design and Assembly below)?
- Which materials will filter large sediments out of the contaminated water?
- Which materials will purify the water?
- What order of materials would provide the most efficient results?
Materials and Equipment
- A computer with access to the internet
- pH strips
- Contaminated water
- Erlenmeyer flask
- Funnel
- Beaker
- Scale
- Alum
- Sodium bicarbonate
- Glass stirring rod
- Sand
- Activated carbon
- Gravel
- Turbidity Sensor
Price List
| Material | Unit | Cost Per Unit |
|---|---|---|
| Sand | 1 tsp | $1.00 |
| Activated Carbon | 1 tsp | $0.50 |
| Gravel | 1 tsp | $0.25 |
Procedure
Note: Water filtered in this experiment SHOULD NOT be consumed
1. Initial Data
- Record all data observed in the lab in the Excel datasheet found here.
- Weigh the empty 500 mL beaker using the scale in terms of grams. Make sure to zero the scale by hitting the tare button before weighing the beaker.
- After you record the weight of the beaker, the TA will add ~200 g of the contaminated water into your beaker. Take note of the color, odor, and any visual observations.
- Your TA will also use a turbidity sensor and measure the initial turbidity of your water sample before and after adding the soil contaminant. Be sure to record the value in your data sheet.
- Reweigh the beaker + contaminated water sample and calculate the weight of just the contaminated water. Record the weight in the datasheet. It should be around 200g.
2. pH Neutralization
Measure the pH of the water before constructing the water filter. The EPA standard for drinking water is a pH from 6.5 to 8.5. Although the human body can occasionally consume liquids with higher and lower pH with no health concerns, excessively drinking acidic water can lead to weakened teeth and gastrointestinal problems.
- Immerse one end of the pH strip into the contaminated water for 10 to 20 seconds.
- Remove the pH strip from the contaminated water after a brief period of time has elapsed.
- Compare the color on the end of the strip to the pH color chart in Figure 4 of this manual page.
- Record the pH and determine if the contaminated water is acidic or alkaline.
- For acidic water, one method of increasing the pH is to add sodium bicarbonate, otherwise known as baking soda, which has a pH of approximately 9. If the solution is acidic, add sodium bicarbonate to it.
- Add in a 1/8 tsp or 0.3g of baking soda to the contaminated water and mix well. To make sure you get the proper amount of baking soda into the water, place the beaker on the scale and hit the tare button. Then slowly add in baking soda until it weighs around 0.3g. Record the data in the datasheet.
- Test the pH of the contaminated water again with a pH strip for 10 to 20 seconds.
- Compare the color on the end of the strip to the pH color chart and record the final pH. If the pH is not between the range of 6.5 to 8.5, repeat step 5 - 8 until the pH meets the EPA standards.
- Weigh the beaker with the contaminated water + baking soda and record the data in the datasheet.
3. Water Filter Design and Assembly
- Add around 0.5 g of alum to the contaminated water. Mix the contaminated water well for 10 seconds. Record the mass of alum added in the datasheet
- Let the contaminated water rest for 10-15 minutes to allow for flocculation to occur. Note any changes observed in the mixture. Your final coagulated & flocculated water sample should look like the one in Figure 11.
- Weigh and record the mass of the contaminated water + alum in the beaker in the datasheet.
- Based on the available materials in Table 1 and a funnel, design a water filter in your lab notes that will most efficiently filter the physical sediments with the following limitations:
- Use a minimum of three materials.
- Each material can only be used a maximum of three times.
- Record the following information in your lab notes:
- Materials used to design the filter
- Order of adding each material
- Quantity of each used material
- Total cost
- Have a TA approve the design.
- Obtain the required materials and assemble the water filter. A gauze pad must be placed on the inside of the funnel to contain the filter materials.
- Weigh an empty Erlenmeyer flask and record the weight in the datasheet.
- Place the assembled water filter in the funnel on top of the Erlenmeyer flask.
- After the 10-15 minute time frame for the flocculation process, slowly pour the contaminated water through the filter. Do not overfill the filter. But before you do, please read the following instructions:
- At this point in the lab you also need to measure the Flow Rate. Have one member in your group start a timer the second the water is poured into the filter. Stop the timer when the water in the Erlenmeyer flask reaches 100mL. Record the time, you will need it to calculate the Flow Rate (6) in Step 18.
- Do not overfill the filter.
- The water must be poured slowly to first filter out all of the clear water. Exclude the floc at the bottom.
- Once that filters through, pour in the rest of the contaminated water sample including the floc.
- Once the contaminated water has been completely filtered, remove the assembled water filter from the Erlenmeyer flask and rest the filter on the now empty beaker.
- Write down any new observations about the filtered water. What color is it? How does its clarity compare to before? Does the water look clean?
- Reweigh the Erlenmeyer flask with the filtered water and calculate the weight of just the filtered water. Record the weight of the flask and the water in the datasheet.
- Have your TA measure the final turbidity of the water. Record the new turbidity reading in the datasheet.
- Calculate the fractional mass lost using (4) and record it in the datasheet. The fractional mass lost normalizes the mass of the physical sediments that is lost from the contaminated water during filtration. A fractional percent mass lost signifies a higher purity of water.
(4)
- Calculate the competition ratio of the filter design using (5). The competition ratio judges how effective the filter is at removing physical sediments from the contaminated water.
(5)
(6)
- Note that 1 = 1 mL,100 represents the 100 mL of water measured earlier in the procedure.
- Share your results with a TA. The TA will record the results in the Competition Spreadsheet.
4. Process Flow Diagram
- Design a simplified industrial water treatment system PFD based on the experiments done in this lab. EG1003 recommends Diagrams.net for creating the PFD. Include:
- pH Neutralization
- Coagulation and flocculation
- Sedimentation
- Filtration
- The design will assume that the initial flow of contaminated water is 1000 kg/s. The contaminated water will flow through the following process units:
- pH Neutralizer
- Mixer
- Filter
- Storage
- Based on the numerical data and the given initial mass flow rate, be sure to add the following:
- Amount of contaminated water entering the system.
- Mass flow rate of alum added to the system.
- Divide the mass of the alum by the mass of the contaminated water to find the flow rate of alum needed for the PFD
- Calculate the mass flow rate of physical sediment removed from the system.
- Calculate the mass flow rate of filtered water leaving the system.
Keep in mind the order of filtration and any other process units needed to logically complete the process flow (for example a mixer after adding the alum). See the PFD example in the Overview section for reference. Be mindful of the mass balance (6).
(6)
The lab work is now complete. Refer to the Assignment section for the instructions to prepare the lab report.
Assignment
Team Lab Report
Follow the lab report guidelines laid out in the EG1004 Writing Style Guide in the Technical Writing section of the manual. Use the outline below to write this report.
- Describe the water treatment process.
- What is pH? Why is it important to maintain the pH of drinking water?
- What potential effects would an increase in turbidity have on a lake?
- What was the final pH of the contaminated water? Is this within the EPA standards for drinking water?
- At what step in the water treatment system was the alum added? What did alum do to the contaminated water? Discuss any observations.
- Discuss minimal design. Describe the importance of minimal design and explain how it was employed in the design. Did the design minimize cost?
- Explain the water filter design, including why certain materials were selected and the order in which they were added.
- Compare your design with the other water filters designed in class. Which was the most cost-effective? Which was the cheapest? Which filtered out the most material?
- Overall, which filter media was the most effective at filtering the contaminated water?
- Explain the thought process behind the PFD designed in Part 3 of the procedure.
- How might the water filter design be improved (both individual and large scale)?
- Include pictures of the water filter, the polluted water, the filtered water, the pH strips, and the complete PFD.
- Discuss what part of the lab each member completed for the group and how it was important to the overall experiment.
Team Presentation
Follow the presentation guidelines laid out in the EG1004 Lab Presentation Format in the Technical Presentations section of the manual. When preparing the presentation, consider the following points.
- Since one term in the competition ratio is cost, present the cost of the water filter. Use the page How to Show Cost Data in Presentations for instructions on how to do this
- How might the water filter design be improved?
- Include pictures of the water filters, the coagulation and flocculation results, the unfiltered water sample, and the filtered water samples
- Which design was the most cost effective and why?
- Include picture/screenshots of the complete PFD
References
References
2018 Edition of the Drinking Water Standards and Health Advisories Tables (2018, March). Retrieved from https://www.epa.gov/sites/production/files/2018-03/documents/dwtable2018.pdf
FAO's Global Water Information System. Prepared by AQUASTAT, Food and Agriculture Organization of the United Nations, www.fao.org/3/I9253EN/i9253en.pdf.
Guppy, L., Anderson, K., 2017. Water Crisis Report. United Nations University Institute for Water, Environment and Health, Hamilton, Canada.
Healthy Water. (2020, March 23). Retrieved July 02, 2020, from https://www.cdc.gov/healthywater Schroeder , Edward D. “Water Resources.” Encyclopedia of Physical Science and Technology, by Robert Allen
Meyers, 3rd ed., Academic Press, 2002, pp. 721–751. https://reader.elsevier.com/reader/sd/pii/B0122274105008218?token=709604225F511CB5F3C8E980E1DB5766BBFF9ABB22B7502BA791BC763A910230D5E1BA5444EE32BC25FD8DF7B67DD75F
Water Treatment. (2015, January 20). Retrieved July 02, 2020, from https://www.cdc.gov/healthywater/drinking/public/water_treatment.html
| ||||||||

![{\displaystyle pH=-log[H^{+}]}](https://wikimedia.org/api/rest_v1/media/math/render/png/085bcfa09edd2b2d817ee499761dc0a43e62817f)
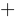 ) and hydroxide ions (OH
) and hydroxide ions (OH ). pH is measured on a scale from 0 to 14. A pH of 7 indicates a neutral solution, which signifies that the concentration of H
). pH is measured on a scale from 0 to 14. A pH of 7 indicates a neutral solution, which signifies that the concentration of H



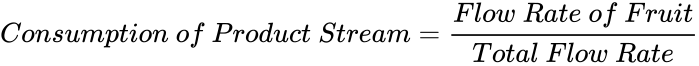
![{\displaystyle Consumption\ of\ Pineapple\ Juice\ Extract={\frac {500\left[{\text{kg/s}}\right]}{675\left[{\text{kg/s}}\right]}}=0.74}](https://wikimedia.org/api/rest_v1/media/math/render/png/b4abdcdde50b0b1e2be3532e4b982d3e22635768)

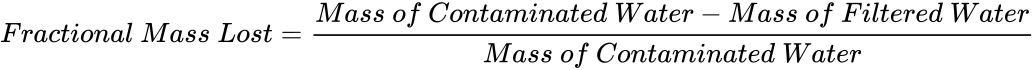
![{\displaystyle Competition\ Ratio={\frac {Fractional\ Mass\ Lost}{Total\ Cost\left[{\$}\right]}}\times FlowRate{\frac {\left[{cm^{3}}\right]}{\left[{s}\right]}}+[{\frac {Filtered\ Water\ Turbidity\left[{V}\right]-Contaminated\ Water\ Turbidity\left[{V}\right]}{Clean\ Water\ Turbidity\left[{V}\right]-Contaminated\ Water\ Turbidity\left[{V}\right]}}]}](https://wikimedia.org/api/rest_v1/media/math/render/png/50983f62db313975e1cee8cc52a51e5414b0fb9d)
![{\displaystyle Flow\ Rate={\frac {100\left[{cm^{3}}\right]}{Filtration\ Time\left[{s}\right]}}}](https://wikimedia.org/api/rest_v1/media/math/render/png/c0f01811638ac099a5c494b80a9e548da5365b68)
 = 1 mL,100
= 1 mL,100